--------------------------------------------------
The "Cicada" Variant: What We Know.
Here is what we know about a new Covid-19 variant in the headlines, nicknamed 'cicada'.
Gage Moreno, 4-1-26
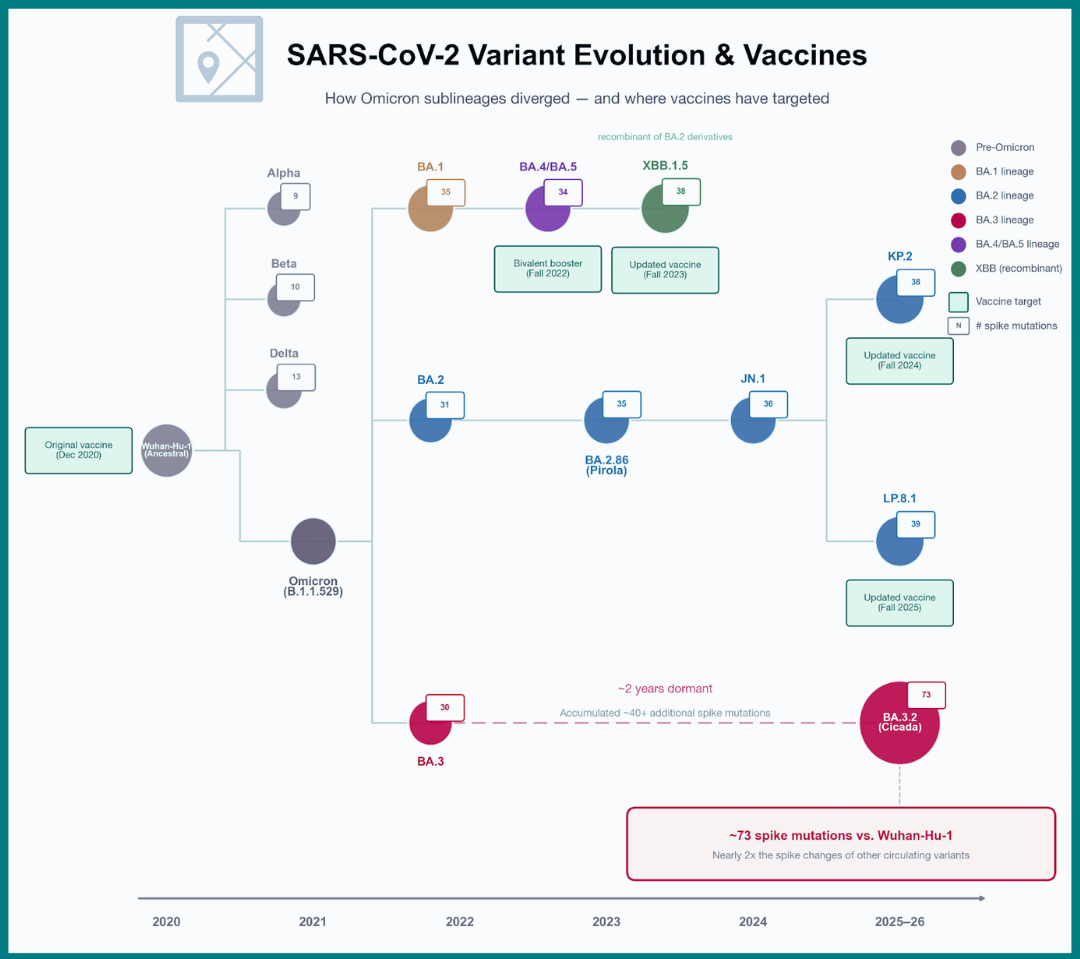
A new Covid-19 variant has been making headlines lately, and this one has an unusual name. BA.3.2, nicknamed "cicada", has been detected in at least 25 U.S. states and 23 countries. Here's what we know so far.
Cicadas are famous for spending years underground before emerging in enormous numbers, sometimes after 13 or 17 years of dormancy. BA.3.2 follows a loosely similar pattern: it was first identified in South Africa in November 2024, circulated at very low levels for over a year largely under the radar, and then emerged more forcefully in Europe last fall before spreading broadly this winter. That pattern of persistence followed by sudden resurgence is what earned it the unofficial nickname.
Most people remember Omicron, the variant that swept through in late 2021 and displaced earlier SARS-CoV-2 strains. BA.3.2 belongs to that same family but has followed a different evolutionary path. Recent Omicron descendants such as JN.1 and LP.8.1–the strains targeted by the current 2025–2026 vaccine–carry on the order of 30–40 mutations in the spike protein. In contrast, BA.3.2 has accumulated roughly 70–75.
That level of divergence is notable, particularly because many of these mutations are concentrated in the spike protein, the part of the virus that binds to human cells and the primary target of neutralizing antibodies. In laboratory studies, the current vaccine appears somewhat less effective at neutralizing or protecting against BA.3.2. However, laboratory experiments do not always translate directly into real-world outcomes.
"BA.3.2 is being closely monitored because its highly divergent spike protein may allow it to partially evade existing immunity from vaccination or prior infection. That could increase the likelihood of reinfection at the population level.
"At the same time, immune evasion does not necessarily mean more severe disease, and the early epidemiologic signals are reassuring. Whether BA.3.2 will drive a substantial wave in the United States remains uncertain, and will depend on both its intrinsic transmissibility and the current immune landscape of the population. But so far, BA.3.2 shows no evidence of higher case numbers or increased severity. WHO assessments have found no evidence of increased hospitalizations, ICU admissions, or deaths associated with the variant.
"Symptoms are similar to other SARS-CoV-2 variants (i.e. cough, fever, fatigue, congestion), and remains susceptible to antivirals like Paxlovid. Laboratory data indicate that BA.3.2 is resistant to several commonly used monoclonal antibodies, including cilgavimab, bebtelovimab, and sotrovimab, but remains sensitive to tixagevimab.
"As of mid-March, BA.3.2 accounted for less than 1% of sequenced cases in the U.S., though wastewater monitoring shows gradual increases.
"This is a practical question where the guidance is clear, even if some of the underlying science is still evolving. The current Covid-19 vaccine formulation, based on recent Omicron-lineage strains (including JN.1 and closely related variants), is expected to remain in place until fall 2026, when an updated version may be introduced. For most healthy adults, a single annual dose is recommended.
"For older adults, immunocompromised individuals, and those with chronic conditions, the recommendations are different. Protection from vaccination declines over time, and these groups both start from a lower peak response and face a higher risk of severe outcomes if infected. For this group, a second dose is recommended at least 6 months after the first.
"We do not yet have definitive real-world data on how well the current vaccine performs specifically against BA.3.2. However, across multiple Omicron waves, a consistent pattern has held: booster doses restored protection against severe disease, even when protection against infection was reduced. That prior evidence is what current recommendations are based on.
"In that context, a second dose spaced roughly six months after the last vaccination is a reasonable way to maintain protection through higher-risk periods. If it has been six months or more since your last dose, discussing a booster with a healthcare provider is a well-supported step, particularly with a variant in circulation that may partially evade existing immunity.
"The broader guidance remains the same. Stay home when sick, use masks in higher-risk indoor settings if appropriate, attend to ventilation, and keep home tests on hand. These tests target conserved regions of the virus and are expected to remain effective for BA.3.2."














.jpg)